Empirical formula worksheets, often available as PDF documents with answers, are crucial tools for mastering stoichiometry. These resources offer focused practice,
strengthening understanding of compositional ratios within chemical compounds and solidifying essential skills.
What is an Empirical Formula?
The empirical formula represents the simplest whole-number ratio of atoms in a chemical compound. Unlike the molecular formula, which shows the actual number of atoms, the empirical formula provides the most reduced ratio. For instance, a compound with a molecular formula of C6H12O6 has an empirical formula of CH2O.
Determining the empirical formula involves calculating the percent composition of each element in the compound, converting those percentages to moles, and then finding the simplest whole-number ratio of moles. Empirical formula worksheets with answers, often in PDF format, guide students through this process. These worksheets typically present percentage compositions, requiring learners to perform calculations to arrive at the correct empirical formula. Understanding this concept is foundational for further studies in chemistry, particularly when analyzing compound composition and relating it to molecular weight.
Why Use Worksheets for Practice?
Empirical formula worksheets are invaluable for solidifying understanding through repetitive practice. Mastering this concept requires applying a multi-step process – calculating percent composition, converting to moles, and simplifying ratios – and worksheets provide structured opportunities to do so. Utilizing resources like empirical formula worksheets with answers (often available as PDF downloads) allows for self-assessment and immediate feedback.
These worksheets move beyond rote memorization, forcing students to actively apply the principles. The availability of answer keys enables independent learning and error identification. Working through various problems builds confidence and reinforces the procedural knowledge needed to tackle more complex stoichiometry challenges. Consistent practice with these materials significantly improves problem-solving skills and prepares students for exams and further scientific study.

Understanding the Basics
Empirical formula worksheets, often in PDF format with answers, require grasping percent composition, mole ratios, and converting grams to moles—fundamental concepts for success.
Calculating Percent Composition
Percent composition is a cornerstone skill when tackling empirical formula worksheets, frequently found as PDFs with answers. It involves determining the mass percentage of each element within a compound. This is achieved by dividing the mass of each element by the compound’s total mass, then multiplying by 100%.
Worksheets often present the compound’s mass or the masses of its constituent elements. Students must then accurately calculate these percentages. Mastering this step is vital, as it directly feeds into determining the mole ratios needed for the empirical formula. Many PDF resources provide step-by-step examples and practice problems to reinforce this concept. Accurate calculations here ensure the subsequent steps, like finding mole ratios, are also correct; Understanding this foundational skill is key to confidently solving empirical formula problems.
Converting Percent to Grams
Once percent composition is established – often practiced via empirical formula worksheets in PDF format with answers – the next step is converting these percentages into grams. This assumes a 100-gram sample of the compound, simplifying the process. Essentially, the percent value directly represents the mass in grams for that element within the 100g sample.
For example, if a compound contains 40% oxygen, this translates to 40 grams of oxygen in a 100-gram sample. Worksheets frequently present scenarios requiring this conversion. This step is crucial because molar mass calculations, essential for determining mole ratios, require mass in grams. PDF practice materials often include exercises specifically designed to build proficiency in this conversion, ensuring students can seamlessly transition to the next stage of empirical formula determination.
Determining Mole Ratios
After converting percent composition to grams – a skill honed through empirical formula worksheets, often found as PDFs with answers – the next critical step is determining the mole ratio of each element. This involves dividing the mass of each element (in grams) by its respective atomic mass obtained from the periodic table. The result is the number of moles of each element present in the compound.
These mole values then form the basis for establishing the simplest whole-number ratio, which defines the empirical formula. Many PDF worksheets provide guided practice, walking students through this process. Understanding this step is paramount, as it bridges the gap between mass percentages and the fundamental building blocks of chemical formulas. Correctly calculating mole ratios is essential for accurate empirical formula determination, and practice materials emphasize this importance.

Solving Empirical Formula Problems
Empirical formula worksheets, frequently in PDF format with answers, provide structured practice. They guide learners through calculating these formulas from percentage compositions and experimental data.
Step-by-Step Problem Solving Approach
Successfully tackling empirical formula worksheets, especially those in PDF format with answers, relies on a systematic approach. First, convert given percentages to grams by assuming a 100g sample. This simplifies calculations.
Next, convert these gram values into moles using the respective atomic masses from the periodic table. This step is fundamental to determining the molar ratios. Then, divide each mole value by the smallest mole value calculated. This yields the simplest whole-number ratio.
If the ratios are not whole numbers, multiply all ratios by a common factor to achieve whole numbers. Finally, write the empirical formula using these whole-number subscripts. Utilizing worksheets with provided solutions allows for self-assessment and error identification throughout this process, reinforcing understanding and building confidence.
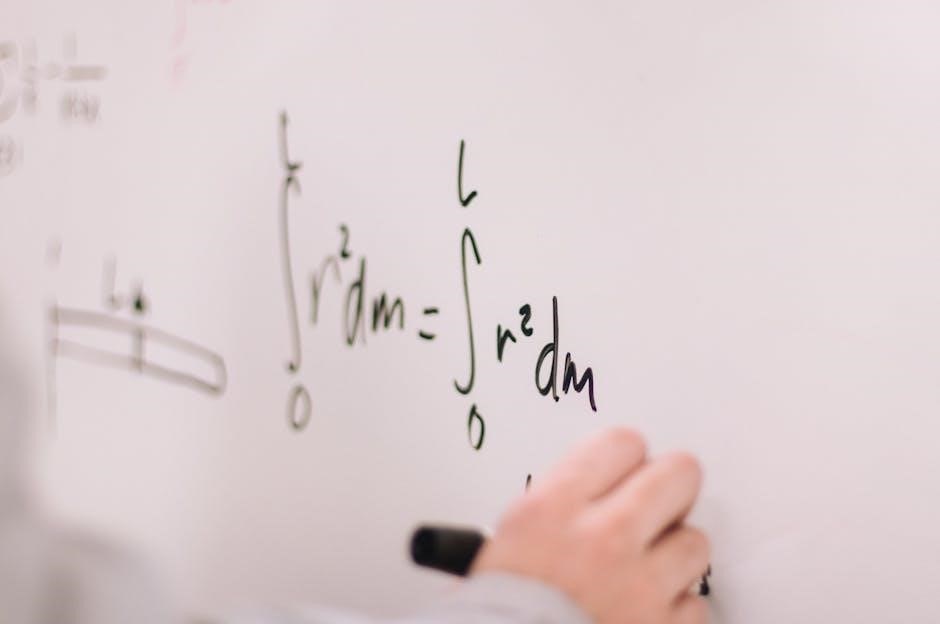
Example Problem 1: Potassium, Phosphorus, and Oxygen
Let’s consider a compound containing 55.3% Potassium (K), 14.6% Phosphorus (P), and 30.1% Oxygen (O). Using a 100g sample, we have 55.3g K, 14.6g P, and 30.1g O.
Convert to moles: K (55.3g / 39.1g/mol = 1.41 mol), P (14.6g / 30.97g/mol = 0.47 mol), and O (30.1g / 16.00g/mol = 1.88 mol). Divide by the smallest mole value (0.47 mol): K (1.41/0.47 = 3.00), P (0.47/0.47 = 1.00), and O (1.88/0.47 = 4.00).
The resulting ratio is K3P1O4. Therefore, the empirical formula is K3PO4. Worksheets, particularly those in PDF format with answers, often present similar problems, allowing students to practice these steps and verify their solutions. Consistent practice is key to mastering empirical formula calculations.
Example Problem 2: Carbon, Hydrogen, and Oxygen
Imagine a compound composed of 40.0% Carbon (C), 6.7% Hydrogen (H), and 53.3% Oxygen (O). Assume a 100g sample: 40.0g C, 6.7g H, and 53.3g O.
Convert to moles: C (40.0g / 12.01g/mol = 3.33 mol), H (6.7g / 1.01g/mol = 6.63 mol), and O (53.3g / 16.00g/mol = 3.33 mol). Divide by the smallest mole value (3.33 mol): C (3.33/3.33 = 1.00), H (6.63/3.33 = 1.99 ≈ 2.00), and O (3.33/3.33 = 1.00).
The resulting ratio is C1H2O1, simplifying to CH2O. Empirical formula worksheets, frequently available as PDF files with answers, provide numerous examples like this. These resources help students develop proficiency in converting percentages to moles and determining the simplest whole-number ratio, a fundamental skill in chemistry. Regular practice using these materials is highly recommended.

Advanced Concepts & Common Challenges
Empirical formula worksheets, including PDF versions with answers, often extend to hydrates and scenarios yielding non-whole number ratios, demanding careful calculation.

Compounds with Hydrates
Hydrates present a unique challenge when determining empirical formulas, as they contain water molecules integrated into their crystalline structure. Empirical formula worksheets, particularly those in PDF format with answers, frequently include problems specifically designed to address these compounds.
The key to solving these problems lies in first determining the mass of the anhydrous salt (the salt without water) and then calculating the mass of water associated with it. This is typically achieved through heating the hydrate to drive off the water, and carefully measuring the mass loss.
Once the masses of both the anhydrous salt and the water are known, they are converted to moles. The mole ratio between the salt and water then reveals the stoichiometric coefficient for water in the hydrate formula. Worksheets often guide students through this process step-by-step, providing practice with these calculations and reinforcing the concept of hydrate stoichiometry. Understanding this process is vital for accurate formula determination.
Dealing with Non-Whole Number Ratios
Occasionally, when calculating mole ratios from percent composition data, you may encounter values that are not whole numbers. This is a common occurrence and doesn’t indicate an error; it simply requires an additional step to arrive at the simplest whole-number ratio representing the empirical formula.
Empirical formula worksheets, especially those available as PDFs with answers, often include examples of this scenario. The solution involves finding the greatest common factor (GCF) of the non-whole number ratios and dividing each ratio by this GCF. This process effectively scales the ratios to the smallest possible whole numbers.
For instance, if you obtain a ratio of 1.5:2.5, the GCF is 0.5. Dividing both values by 0.5 yields 3:5, representing the final, simplified empirical formula ratio. Worksheets provide ample practice in identifying the GCF and applying this scaling technique, ensuring students can confidently handle these situations.

Resources & Practice Materials
Numerous online platforms and PDFs offer empirical formula worksheets with answers, providing extensive practice. These materials are invaluable for skill development and assessment.
Where to Find Empirical Formula Worksheets with Answers (PDF)
Locating reliable empirical formula worksheets with answers in PDF format is straightforward. Several educational websites specialize in chemistry resources, offering downloadable practice sets. Sites like ChemWorksheets.com and ScienceSpot provide a variety of worksheets catering to different skill levels, from introductory to advanced.
Many teachers also create and share their own worksheets, often available through school learning management systems or personal websites. A quick Google search using keywords like “empirical formula practice problems PDF” or “stoichiometry worksheet with key” will yield numerous results.
Furthermore, platforms like Khan Academy offer interactive exercises and video tutorials that complement worksheet practice. When selecting a worksheet, ensure it includes a detailed answer key to facilitate self-assessment and error identification. Look for worksheets that present a range of problem types, including those involving percent composition, mole ratios, and hydrate calculations, to ensure comprehensive practice.
Online Empirical Formula Calculators
While empirical formula worksheets with answers (PDF) provide structured practice, several online calculators can assist in verifying solutions and understanding the process. Websites like Vedantu and CalculatorSoup offer free empirical formula calculators where users input element masses or percent compositions. These tools instantly determine the empirical formula, serving as a valuable check against hand-calculated results.
These calculators aren’t meant to replace practice, but rather to reinforce learning and identify areas where students might struggle. They can quickly handle complex calculations, allowing users to focus on the underlying concepts.
Remember to understand how the calculator arrives at the answer, not just accepting the result blindly. Combining worksheet practice with calculator verification offers a powerful learning strategy, ensuring both computational accuracy and conceptual understanding of empirical formula determination.
Key Terms & Definitions (Empirical vs. Molecular Formula)
Successfully navigating empirical formula worksheets with answers (PDF) requires a firm grasp of key terminology. The empirical formula represents the simplest whole-number ratio of atoms in a compound – for example, CH2O. Conversely, the molecular formula shows the actual number of atoms of each element in a molecule, like C6H12O6.
A compound’s molecular formula is always a whole-number multiple of its empirical formula. Understanding this relationship is vital when solving problems. The empirical formula simplifies the composition, while the molecular formula reveals the true structure.
Distinguishing between these two formulas is crucial for interpreting results from worksheets and calculators. Mastering these definitions ensures a solid foundation for further study in stoichiometry and chemical formulas.

Answer Key Considerations
Answer keys for empirical formula worksheets with answers (PDF) should be carefully reviewed. Verify calculations and understand why an answer is correct, not just that it is.
Checking Your Work & Identifying Errors
When utilizing an empirical formula worksheet with answers (PDF), meticulous self-assessment is paramount. Begin by verifying each step of your calculations – percent composition, gram conversions, and mole ratio determinations. Ensure accurate atomic masses are used, and double-check any division or multiplication.
Common errors include incorrectly converting percentages to grams, miscalculating mole ratios, or prematurely rounding numbers, leading to inaccurate simplified formulas. If your empirical formula differs from the answer key, systematically retrace your steps.
Pay close attention to the smallest whole-number ratio; this is the defining characteristic of an empirical formula. Consider if you’ve correctly identified the limiting reactant or if stoichiometric coefficients were applied appropriately. Utilizing the provided answer key isn’t merely about finding the correct answer, but understanding the process to avoid repeating mistakes.
Understanding the Significance of the Empirical Formula
Successfully completing an empirical formula worksheet with answers (PDF) reveals more than just calculation skills; it illuminates the core representation of a compound’s elemental composition. The empirical formula signifies the simplest whole-number ratio of atoms in a substance, offering fundamental insight into its building blocks.
While the molecular formula displays the actual number of atoms, the empirical formula provides a standardized way to compare different compounds. It’s crucial for identifying unknown substances and understanding their basic chemical nature.
Mastering this concept, through practice with worksheets, is essential for progressing to more complex stoichiometry problems. Recognizing that a compound can have the same empirical and molecular formula, or a molecular formula that’s a multiple of the empirical formula, demonstrates a deeper comprehension of chemical formulas and their implications.
